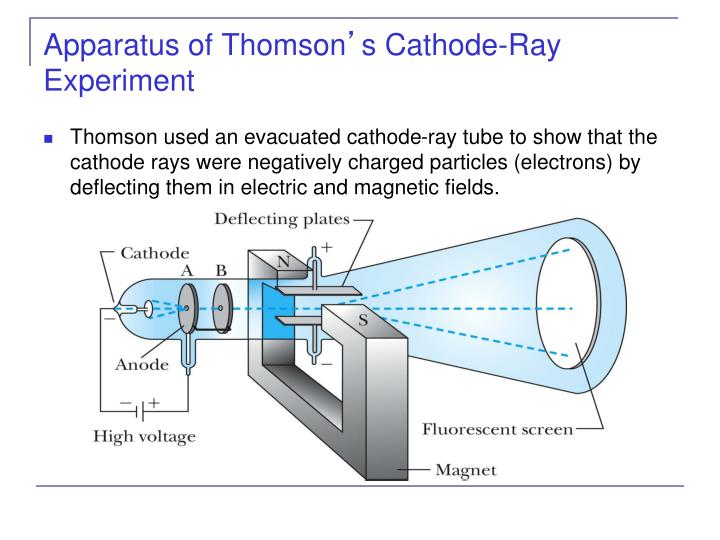
In the “plum pudding” model of the atom, negatively charged corpuscles were like raisins suspended in a positively charged cake, resulting in a neutral atom.įor his experiments, Thomson relied on a specialized vacuum tube known as a Crookes tube (more about Crookes and his tubes in a bit), in which he observed and photographed various phenomena, including the effect of a magnetic force on the electrical discharge at high pressure. This did not fit neatly within the Classical Worldview, which held that everything was composed of immutable and indivisible atoms. 
French and British scientists, meanwhile, were beginning to argue that cathode rays were electrified subatomic particles. Ether is part of what historians call the Classical Worldview, a 19th-century synthesis of physics that has its roots in Aristotle and Newton. Many German physicists believed that visible cathode rays resulted from an interaction with the ether-a colorless, weightless substance that enveloped all of space. The purely scientific question-What are cathode rays?-was also wrapped up in national identity. But that knowledge was hard won, after decades of investigations by many players. We now know these rays are streams of electrons emerging from the cathode (or negative electrode) of a vacuum tube. 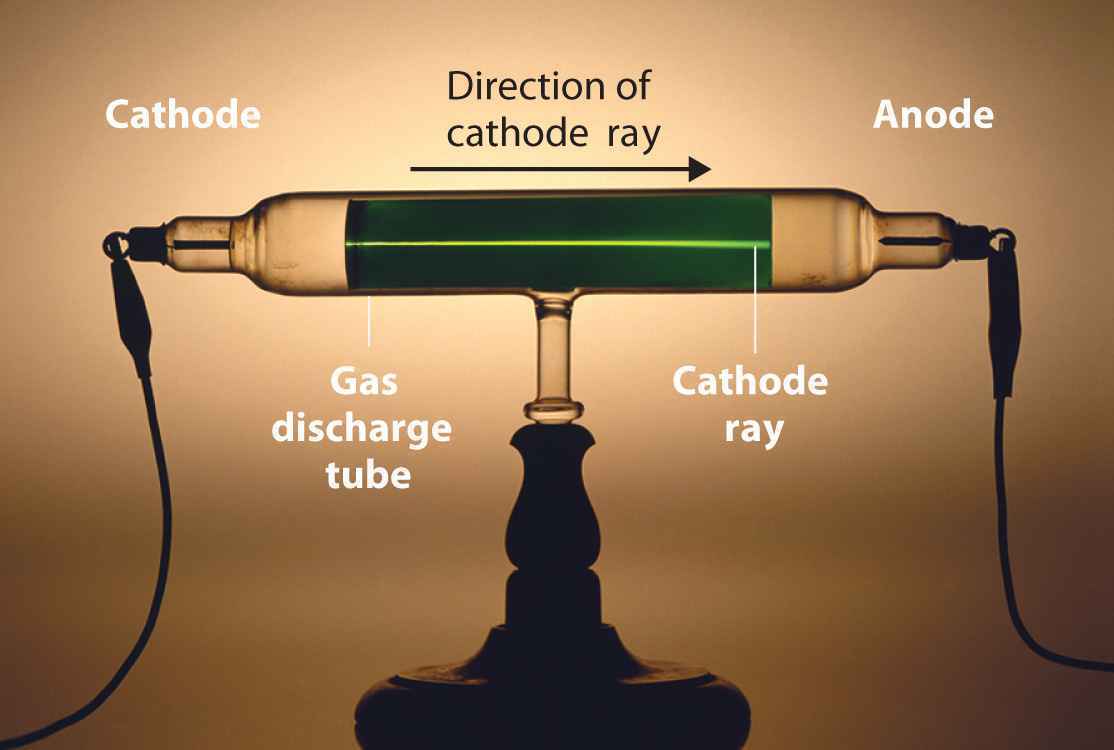
Much of Thomson’s research was devoted to understanding the nature of cathode rays. In 1884, he was appointed the Cavendish Professor of Experimental Physics and began his lifelong study of electromagnetism. In 1876, Thomson had received a scholarship to study at the University of Cambridge’s Trinity College, and four years later he graduated with a degree in mathematics. Thomson was a key player in establishing this new direction in physics. Little did scientists know that atomic theory was about to upend science and change our fundamental understanding of matter. Electricity was being tamed, and theories of thermodynamics were coalescing to explain the workings of steam engines. Thomson gave a lecture detailing his and others’ experiments with the energetic beams inside cathode-ray tubes.Īs the 19th century was coming to a close, many prominent thinkers believed that all of the great discoveries in science had already been made. The more I researched, the more I contemplated what it meant to discover something.Ĭathode rays, vacuum tubes, and the birth of atomic theoryĮxactly 125 years ago, the British physicist J.J. Many people, in fact, had a reasonable claim to aspects of the electron’s discovery. The challenge then was to find a museum artifact that captured that discovery.Ī Braun vacuum tube, like the one pictured at top, seemed like a good choice, because its inventor, Karl Ferdinand Braun, created it to study beams of electrons, and Thomson used a similar instrument for his experiments.īut as I dug into the histories of Thomson and Braun, I learned that theirs were two parallel stories involving many of the same players and similar outcomes (both men won a Nobel Prize in Physics), but having little else in common. For this month’s column, I knew that I wanted to write about the 125th anniversary of the electron’s discovery, which for simplicity’s sake I pegged to Thomson’s lecture. Of course, history is always more muddled than that. Thomson is often hailed as the discoverer of the electron based on that lecture 125 years ago. Corpuscles are electrons, and the plum pudding model gave way to Ernest Rutherford’s nuclear model in 1911. Thomson, who merely endorsed the idea.Ĭorpuscles and pudding are not how we think about the structure of an atom today. The model also became known as the Thomson model, although its chief proponent was William Thomson (Lord Kelvin), not J.J. In Thomson’s analogy, negatively charged corpuscles were like raisins suspended in a positively charged cake, resulting in a neutral atom. This model of the atom became known as the “plum pudding” model, so named for the popular English dessert. Thomson described his experiments with cathode rays to verify the existence of these subatomic corpuscles. “The atoms of the ordinary elements are made up of corpuscles and holes, the holes being predominant,” he continued. Thomson, during a lecture at the Royal Institution in London, on 30 April 1897. What should my next move be? The project ran properly before the Netbeans upgrade, but not after, and no code was changed.“We shall call such particles corpuscles,” announced the physicist J.J. I’m guessing this has something to do with flatpak being sandboxed, but I’m still not sure if even that is the issue. I changed the Java platform from JDK19, to JDK20, however, the Netbeans info center seems to report java 11, despite my java -version being: java version "20". This caused an error in one of the projects I’m working on, and for some reason, in the media player (I suspect some backend issue, not a code issue).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
